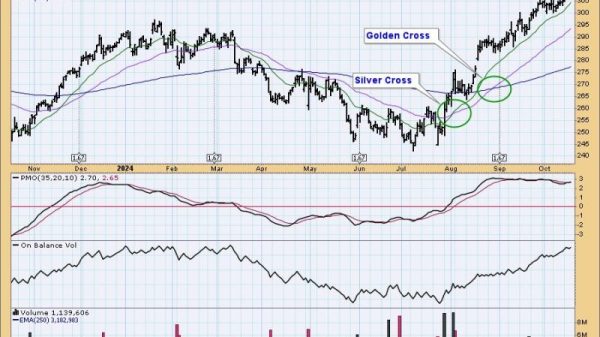
Copper Refining Process: Mining, Extraction, and Refining
The process of refining copper begins at the mining stage, where copper ore is extracted from the earth’s crust. These resources contain varying concentrations of copper and are either mined using open pit or underground methods.
Open-pit mining is typically used when the copper deposit is near the surface and has a higher grade. The ore is extracted, loaded onto trucks, and transported to a processing facility. Underground mining, on the other hand, is employed when the deposit is deep within the earth. This process requires the sinking of shafts and driving of tunnels to reach the copper deposit.
Once the copper ore is harvested, it moves to the next stage, the extraction process. The extraction process, known as beneficiation, involves several steps, including crushing, grinding, and concentration. Here, the ore is crushed and ground into a powder then mixed with water to create a slurry. This slurry undergoes a process called flotation, wherein the copper minerals attach to air bubbles and rise to the surface, forming a froth that is skimmed off for further processing. This concentrate, usually 25 to 35% copper, is then ready for smelting.
The smelting stage involves heating the copper concentrate in furnaces to remove impurities and unwanted elements and to produce matte, a mixture of copper, iron, and sulfide. This product, which contains between 45 to 70% copper, is further refined in a converter, where it undergoes multiple burnings to eliminate iron sulfur and other impurities to produce blister copper, which contains about 98.5-99.5% copper.
The last stage is the electrowinning and electrorefining process, which purifies the blister copper to create copper of high purity, generally over 99.9%. In electrowinning, the copper ions are reduced to copper metal at the cathode while the anode erodes. This copper is now ready for market use.
Electrorefining, a similar process, is used when refining an even purer grade of copper. In this process, the blister copper is used as a positive anode and a pure copper sheet acts as a negative cathode. When electric current is passed, copper ions migrate from the anode to the cathode. The result is a sheet of extremely pure copper, often used in highly sensitive applications, such as electronic components.
One cannot discuss copper refining without addressing the environmental